The carbide of tantalum, when combined with graphite, forms a hard composite that is coated on the cutting edges of high-speed machine tools. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in H2SO4: Molar Mass (g/mol) H (Hydrogen) 2 × 1.00794 2.01588. 
Zinc is found in the minerals zinc blende (sphalerite) (ZnS), calamine, franklinite. It is a bluish-silver, ductile metal that reacts with alkalis and acids. The origin of the name comes from the German word zink. The oxide of tantalum is used to increase the refractive index of camera lens. Relative atomic mass: 65.38 (2) Zinc has been known since ancient times.Another application of Ta is in the making of jewelry and accessories like rings and wristbands.The element is used in the fabrication of chemical process equipment, nuclear reactors, and aircraft and missile parts. Calculate the molar mass of C6H8O7 in grams per mole or search for a chemical formula or substance.It finds application as a component in AC/DC rectifiers.Another use is as an economical substitute for platinum. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021.It is often used as an electrode in neon lights.Tantalum increases the strength, ductility, and melting point of alloys used in turbine blades, and rocket nozzles and nose caps of supersonic aircraft.In fact, some clinical trials have also suggested the use of porous tantalum as a safe orthopedic and dental implant As the metal causes no immune response when in contact with bodily fluids, it offers some medical uses such as making orthopedic joint implants, surgical skull plates, suture materials, and woven gauze (mesh).The high corrosion resistance of the element makes it an important component in large-scale equipment such as pipes, tubing, tanks, heat exchangers, and vessels exposed to extreme corrosive materials.The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. Due to this reason, it is suitable for portable electronic devices like mobile phones. Prepare a concept map and use the proper conversion factor. A thin layer of Ta is also coated on other metals to achieve a higher capacitance in a smaller volume. Tantalum is used in capacitors where an oxide layer formed on it acts as an insulator (dielectric) to store more charge. 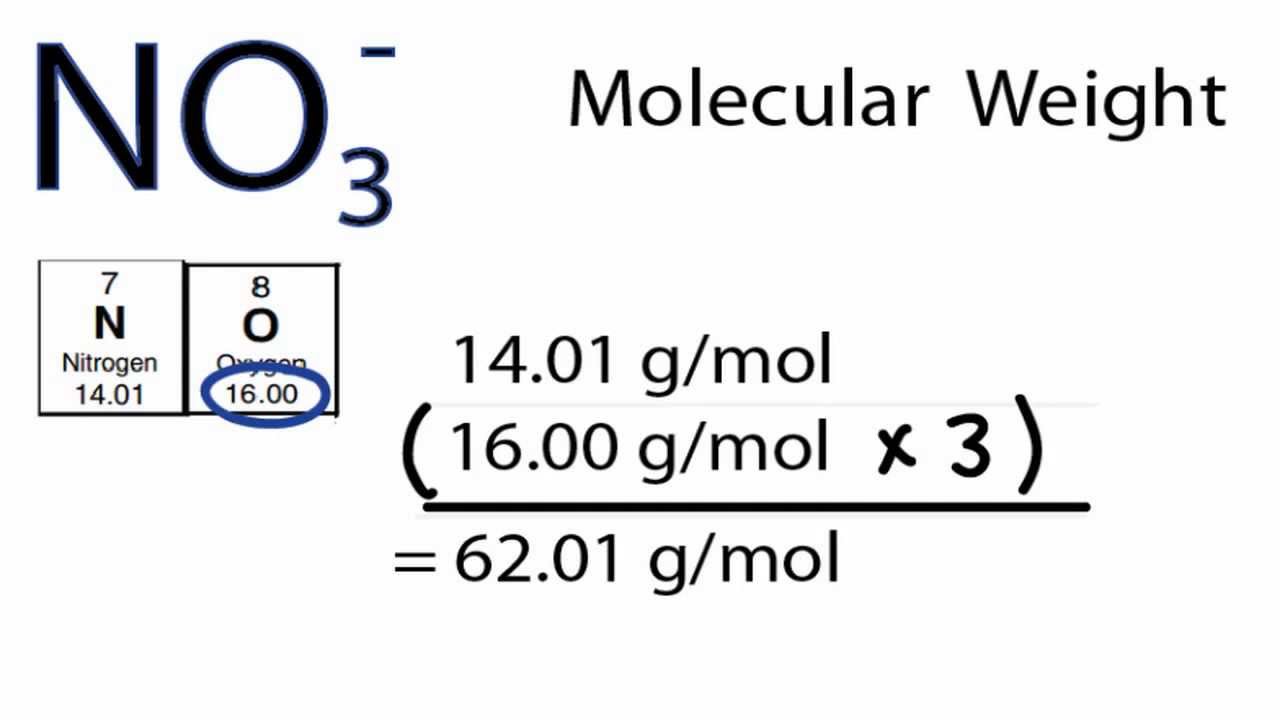
For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. Visualize trends, 3D orbitals, isotopes, and mix compounds. Use this visual tool for calculating molar mass for any chemical formula. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. Interactive periodic table showing names, electrons, and oxidation states. Before placing in container D, check the pH with pH universal indicator strips (Cat. The concept of molar mass can also be applied to compounds. Continue rinsing.ĨB Non-combustible, corrosive hazardous materialsġ3 Bases and alcoholates should be diluted if necessary by carefully stirring them into water and then neutralized (protective gloves, fume cupboard!) with hydrochloric acid (Cat. using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass). Remove contact lenses, if present and easy to do. The molar mass and molecular weight of Cu(CH3COO)2 (Copper(Ii) Acetate) is 181.634. P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. 
This periodic table is suitable for a computer and mobile device wallpaper. The IUPAC doesnt update these values annually, so these are the most recent values for 2019. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the. P303 + P361 + P353: IF ON SKIN (or hair): Take off immediately all contaminated clothing. This color periodic table contains the accepted standard atomic weights (atomic masses) of each element as accepted by the IUPAC. Calculate the molar mass of Titanium in grams per mole or search for a chemical formula or substance. P301 + P312: IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. P280: Wear protective gloves/ protective clothing/ eye protection/ face protection/ hearing protection. This compound is also known as Tripotassium Phosphate. H314: Causes severe skin burns and eye damage. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
